Molecular Self‐Doping Controls Luminescence of Pure Organic Single Crystals
Olga D. Parashchuk, Artur A. Mannanov, Vladislav G. Konstantinov, Dmitry I. Dominskiy, Nikolay M. Surin, Oleg V. Borshchev, Sergei A. Ponomarenko, Maxim S. Pshenichnikov, Dmitry Yu. Paraschuk
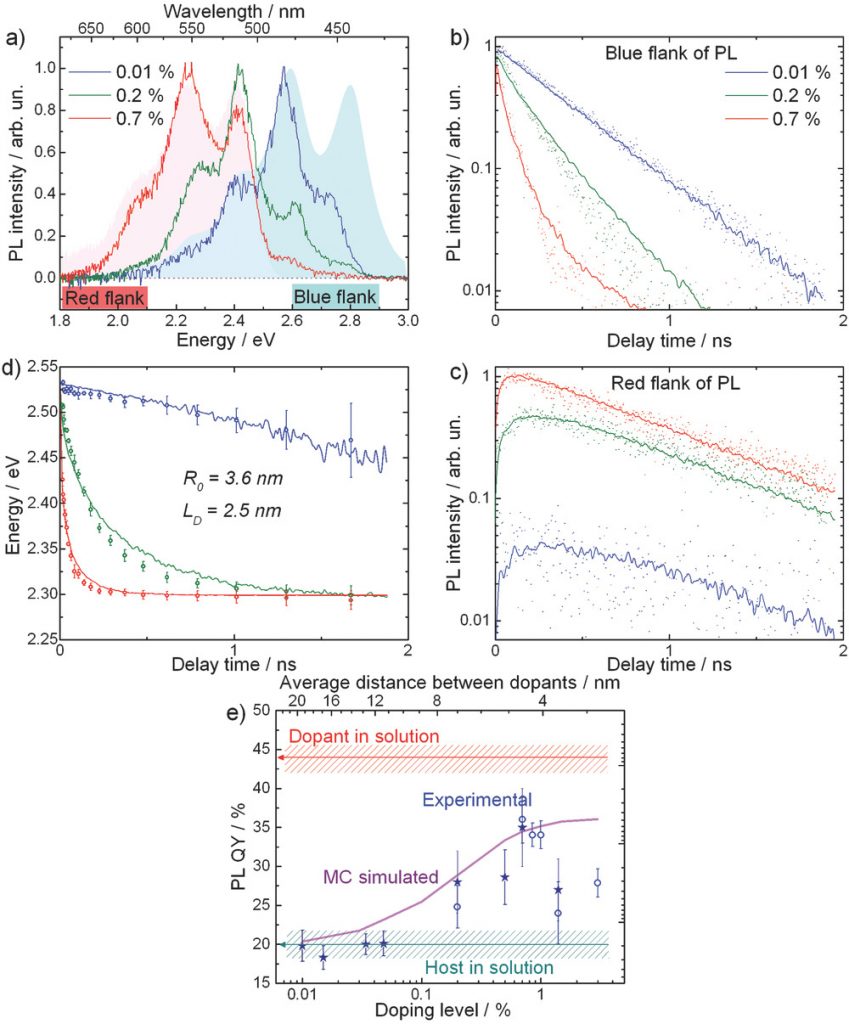
Organic optoelectronics calls for materials combining bright luminescence and efficient charge transport. The former is readily achieved in isolated molecules, while the latter requires strong molecular aggregation, which usually quenches luminescence. This hurdle is generally resolved by doping the host material with highly luminescent molecules collecting the excitation energy from the host. Here, a novel concept of molecular self‐doping is introduced in which a higher luminescent dopant emerges as a minute‐amount byproduct during the host material synthesis. As a one‐stage process, self‐doping is more advantageous than widely used external doping. The concept is proved on thiophene–phenylene cooligomers (TPCO) consisting of four (host) and six (dopant) conjugated rings. It is shown that <1% self‐doping doubles the photoluminescence in the TPCO single crystals, while not affecting much their charge transport properties. The Monte‐Carlo modeling of photoluminescence dynamics reveals that host–dopant energy transfer is controlled by both excitonic transport in the host and host–dopant Förster resonant energy transfer. The self‐doping concept is further broadened to a variety of conjugated oligomers synthesized via Suzuki, Kumada, and Stille crosscoupling reactions. It is concluded that self‐doping combined with improved excitonic transport and host–dopant energy transfer is a promising route to highly luminescent semiconducting organic single crystals for optoelectronics.
